The Elusive Engram: An Experiment in Memory Implantation
The human hippocampus, considered a key area for memory formation. Immunocytochemical staining for calcium-binding proteins is used in this horizontal section to differentiate major areas. Source – http://vida.neurocure.de/research_interests.html
With each passing minute, multitudes of memories surge through our minds as we recollect past experiences and encode new ones. Since the dawn of introspective thought, humans have wondered where such experiences might be encoded, if a physical encoding is even possible [1]. Fast forward to the 20th century and we now have pioneering works from the likes of Carl Lashley, acclaimed for his application of the term “engram” to describe a physical location and mechanism by which a specific memory is encoded, a memory trace so to speak [3]. Lashley’s theories have proved highly influential (though his rat lesion methodologies have been criticized [2]), inspiring countless other neuroscientists such as Richard F. Thompson [6] and Howard Eichembaum [7] to embark on the quest to find the engram.
Fast forward to the 21st century and we continue to see groundbreaking work in engram research. A most recent study published in July 2013 titled “Creating a False Memory in the Hippocampus” [5] provides strong evidence for a functional memory engram through a novel memory implantation procedure. The study was conducted by the Susumu Tonegawa’s RIKEN-MIT Lab, which seeks to uncover the neural mechanisms underlying learning and memory. In this experiment, Tonegawa’s team of neuroscientists were able to implant artificial memories into the brains of mice using optogenetics, a technology in which the activity of specific neurons can be modulated by exposure to certain wavelengths of light. The specific memory manipulated in this study was a conditioned fear response to a mild electrical foot shock.
The methods of this study builds off the findings of a previous publication by Tonegawa’s team from March 2012 [4], where they identified a population of dentate gyrus (DG) hippocampal cells that when optogenetically stimulated, can activate behavioral recall of a context-dependent fear memory; the team named these cells “contextual memory-engram cells”. In the 2013 study, these hippocampal cells were made susceptible to optogenetic techniques by transforming them to express channelrhodopsin-2 (ChR2), a protein that induces excitation in a cell when exposed to (in this case) blue light. The expression and resultant labeling of hippocampal cells with ChR2 was experimentally induced by the absence of doxycycline (Dox) in the diet of the mice, where ChR2 is only expressed when mice do not consume Dox (off Dox condition). ChR2 was also made to be produced whenever c-fos, a gene expressed in response to a novel environment by hippocampal neurons [8], is expressed.

An example of optogenetics in a rat: specific wavelengths of light can be used to control genetically modified neurons. Source – https://a248.e.akamai.net/f/574/7105/8d/www.extremetech.com/wp-content/uploads/2013/01/optogenetics-640×353.jpeg
With these modifications, the mice were allowed to explore Room A on Day 1 while in the off Dox condition. This induced ChR2 labeling of the DG neurons activated during the exploration. The mice also behaved normally in this situation. Later, the mice were surgically modified with an apparatus that can deliver blue light to the ChR2 tagged neurons at the researcher’s discretion. On Day 2, while on Dox, the post-surgery mice explored Room B, a distinctly different room, where they received foot shocks. Unlike in Room A, these mice elicited a fear response to the shocks. The shocks were also accompanied by blue light administered to the ChR2 tagged neurons, thus activating these neurons. Since some of these tagged neurons were specific only for Room A, the researchers hypothesized that the fear response became encoded not only for Room B, but Room A as well.
The researchers tested this hypothesis by bringing the mice back into Room A on Day 3. As expected, the mice exhibited a fear response similar to the one they showed in Room B. To validate these results, a control group was subjected to the same procedures except that they did not receive any blue light stimulation. The researchers were unable to induce a fear response in Room A for this control. Furthermore, both the experimental and control groups were exposed to another distinct Room C. The control group again did not show a fear response while the experimental group showed only low levels of fear response, suggesting a degree of overlap in neurons that encode for Room A and C. Finally, c-fos expression was measured in the basolateral amygdala (BLA), an area associated with fear response that receives information from the hippocampus. Fear responses in both Room A and B elevated c-fos expression in the BLA, suggesting that the artificial fear response in room A is genuine.
The experimental procedures were repeated for mice that were ChR2 labeled for hippocampal area CA1 instead of DG. However, in this condition the researchers were unable to produce a fear response in either Room A or C using these neurons. They suggest that this might be because CA1 neurons have a higher degree of overlap in neurons encoding for different rooms than DG neurons.
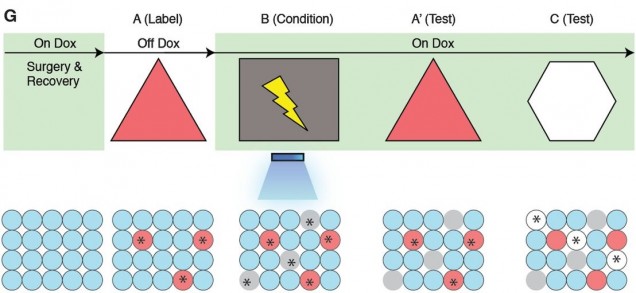
Graphical representation of the procedure. The light green shading indicates the presence of Dox in the diet. Prime indicates a revisited room. The yellow lightning symbol and blue shower symbol indicate foot shocks and blue light delivery, respectively. Red circles represent neurons encoding for Room A that are labeled with ChR2. Gray and white circles represent neurons encoding Rooms B and C, respectively. Asterisks indicate neurons activated either by exposure to a novel room or blue light stimulation. Source – [5]
In summary, Tonegawa’s group concluded that they were able to implant a false memory, in the form of a fear response, into dentate gyrus hippocampal neurons. These findings present a major step forward in the area of memory research and provides significant evidence in support for memory engrams. In fact, Howard Eichenbaum, director of Boston University’s Center for Memory and Brain, has responded to Tonegawa’s findings saying that, “They identified a neural network associated with experience in an environment, attached a fear association with it, then reactivated the network to show that it supports memory expression. That, to me, shows for the first time a true functional engram” [7]. In their 2013 paper, Tonegawa’s team goes on to expresses that, “Our experiments provide an animal model in which false and genuine memories can be investigated at the memory-engram level…In humans, memory distortions and illusions occur frequently…[we] speculate that the formation of at least some false memories in humans may occur in natural settings through the internally driven retrieval of a previously formed memory and its association with concurrent external stimuli of high valence”. Susumu Tonegawa’s RIKEN-MIT Lab plans to conduct further studies on memory distortions and hopefully uncover more truths about the elusive engram [7].
~ Emir Turkes
Sources:
[1] Heil, J. (2004). Minds and Bodies as Distinct Substances. In Philosophy of mind: A guide and anthology (pp. 36-50). Oxford: Oxford University Press.
[2] Hübener, M., & Bonhoeffer, T. (2010). Searching for Engrams. Neuron,67(3), 363–371-363–371. Retrieved November 23, 2013, from http://www.sciencedirect.com/science/article/pii/S0896627310005179#bib58
[3] Lashley, K. (1950). In Search of the Engram. Society of Experimental Biology, Symposium 4, 454–482. Retrieved November 23, 2013, from http://gureckislab.org/courses/fall13/learnmem/papers/Lashley1950.pdf
[4] Liu, X., Ramirez, S., Pang, P., Puryear, C., Govindarajan, A., Deisseroth, K., & Tonegawa, S. (2012). Optogenetic stimulation of a hippocampal engram activates fear memory recall. Nature, 484, 381–385-381–385. Retrieved November 23, 2013, from http://www.nature.com/nature/journal/v484/n7394/full/nature11028.html
[5] Ramirez, S., Liu, X., Lin, P., Suh, J., Pignatelli, M., Redondo, R., . . . Tonegawa, S. (2013). Creating a False Memory in the Hippocampus. Science, 341(6144), 387-391. Retrieved November 23, 2013, from http://www.sciencemag.org/content/341/6144/387.full?sid=c938c93c-1ea9-4a3d-a351-d30d9752fe3d
[6] Thompson, R. (1976). The search for the engram. American Psychologist, 31(3), American Psychologist-American Psychologist. Retrieved November 23, 2013, from http://psycnet.apa.org/journals/amp/31/3/209/
[7] Trafton, A. (2013, July 25). Neuroscientists plant false memories in the brain. Retrieved November 23, 2013, from http://newsoffice.mit.edu/2013/neuroscientists-plant-false-memories-in-the-brain-0725
[8] VanElzakker, M., Fevurly, R., Breindel, T., & L. Spencer, R. (2008). Environmental novelty is associated with a selective increase in Fos expression in the output elements of the hippocampal formation and the perirhinal cortex. Learn Mem, 15(12), 899–908-899–908. Retrieved November 23, 2013, from https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2632843/
March 10, 2014
Quite interesting experiment! I didn’t know it was possible to implant false memories into mice. I wonder how much mice actually remember. How big is their memory?