Our Research
Oral Squamous Cell Carcinoma
Lysine-specific demethylase 1 controls key OSCC preneoplasia inducer STAT3 through CDK7 phosphorylation during oncogenic progression and immunosuppression. Article Link

Oral squamous cell carcinoma (OSCC) progresses from preneoplastic precursors via genetic and epigenetic alterations. Previous studies have focused on the treatment of terminally developed OSCC. However, the role of epigenetic regulators as therapeutic targets during the transition from preneoplastic precursors to OSCC has not been well studied. Our study identified lysine-specific demethylase 1 (LSD1) as a crucial promoter of OSCC, demonstrating that its knockout or pharmacological inhibition in mice reversed OSCC preneoplasia. LSD1 inhibition by SP2509 disrupted cell cycle, reduced immunosuppression, and enhanced CD4+ and CD8+ T-cell infiltration. In a feline model of spontaneous OSCC, a clinical LSD1 inhibitor (Seclidemstat or SP2577) was found to be safe and effectively inhibit the STAT3 network. Mechanistic studies revealed that LSD1 drives OSCC progression through STAT3 signaling, which is regulated by phosphorylation of the cell cycle mediator CDK7 and immunosuppressive CTLA4. Notably, LSD1 inhibition reduced the phosphorylation of CDK7 at Tyr170 and eIF4B at Ser422, offering insights into a novel mechanism by which LSD1 regulates the preneoplastic-to-OSCC transition. This study provides a deeper understanding of OSCC progression and highlights LSD1 as a potential therapeutic target for controlling OSCC progression from preneoplastic lesions.
——————————————————————————————————————————–
LSD1 inhibition corrects dysregulated MHC-I and dendritic cells activation through IFNγ-CXCL9-CXCR3 axis to promote antitumor immunity in HNSCC. Article Link

Poor infiltration of CD8+ T cells and dysregulated MHC-I confer resistance to anticancer clinical therapies. This study aimed to elucidate the mechanisms of lysine-specific demethylase 1 (LSD1, encoded by KDM1A gene) in antitumor immunity in Head and Neck Squamous cell carcinoma (HNSCC). LSD1 inhibition in syngeneic and chronic tobacco carcinogen-induced HNSCC mice recruited activated dendritic cells (DCs), CD4+ and CD8+ T cells, enriched interferon-gamma (IFNγ) in T cells, CXCL9 in DCs, and CXCR3 in T cells, as evaluated using flow cytometry and single cell RNA-seq analysis. Humanized HNSCC mice and TCGA data validated the inverse correlation of KDM1A with DC markers, CD8+ T cells, and their activating chemokines. Kdm1a knockout in mouse HNSCC and LSD1 inhibitor treatment to co-culture of human HNSCC cells with human peripheral blood mononuclear cells (PBMCs) resulted in MHC-I upregulation in cancer cells for efficient antigen presentation in tumors. Overall, LSD1 inhibition in tumor cells upregulates MHC class I and induces DCs to produce CXCL9, which in turn activates CD8+ T cells through the CXCL9-CXCR3 axis to produce IFNγ. Finally, we identified a novel mechanism by which LSD1 inhibition promotes the activation of H3K4me2 and its direct interaction with MHC-I to induce antitumor immunity. This may have implications in poorly immunogenic and immunotherapy-resistant cancers.
Osteoarthritis

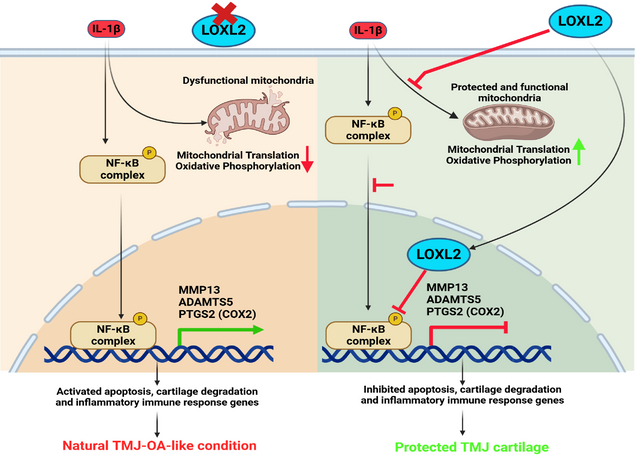
Temporomandibular joint osteoarthritis (TMJ-OA) affects a significant proportion of the population worldwide. However, there has been no substantial progress in the development of FDA-approved drugs for treatment due to a lack of understanding of the specific factors regulating key TMJ-OA molecular mechanisms. Lysyl Oxidase Like-2 (LOXL2) promotes knee joint cartilage protection, and it is downregulated in TMJ-OA animal model. We evaluated the role of LOXL2 in TMJ cartilage, its molecular mechanism and gene networks using in vivo Loxl2 knockout mice (Acan-Cre; Loxl2flox/flox) and ex vivo goat TMJ cartilage. Our results show that Loxl2 knockout in mice cartilage upregulates Il1b, Mmp9, Mmp13, Adamts4, and Adamts5, whereas it reduces the levels of aggrecan and proteoglycan. Loxl2 deleted TMJ cartilage show a higher enrichment of inflammatory response, TNFA signaling via NFKB, extracellular matrix (ECM), and collagen degradation pathway network. Conversely, LOXL2 treatment reduces interleukin-1 beta (IL-1β)-induced expression of Mmp13, protects mitochondrial function and ECM from degeneration. Importantly, LOXL2 attenuates IL-1β-induced chondrocyte apoptosis via phosphorylation of NF-κβ and expression of pain-related gene PTGS2 (COX2). Taken together, Loxl2 knockout mice exacerbate TMJ-OA through cartilage/ECM degradation, mitochondrial dysfunction, chondrocyte apoptosis, and inflammatory gene expression, whereas LOXL2 treatment mitigates these effects.
——————————————————————————————————————————–
LOXL2 alleviates post-traumatic knee osteoarthritis and pain. Article Link

Cartilage has limited potential for self-regeneration, and damage can lead to structural, molecular, and functional aberrations, leading to osteoarthritis (OA). Traumatic knee injuries can also lead to cartilage degeneration and post-traumatic OA (PTOA). This study aimed to explore whether lysyl oxidase-like 2 (LOXL2) deletion promotes PTOA-induced transcriptional changes similar to those in human OA, as well as the upregulation of collagen degradation, inflammation, and pain-related gene networks. LOXL2 was found to be downregulated in mouse knee PTOA. Aggrecan promotes specific deletion of Loxl2 in knee cartilage, shows OA-like molecular changes, and aggravates mouse PTOA. Furthermore, transcriptional analysis revealed the upregulation of cartilage degeneration factors, signatures of inflammatory M1 macrophages, and pain. These Loxl2 deleted PTOA mice have a molecular resemblance to the human knee OA pathogenic gene signature, which could lead to OA and pain. Interestingly, intra-articular injection of adenovirus-delivered LOXL2 rescued knee joint function, alleviated cartilage degeneration, restored treadmill-running capabilities, and reduced mechanical allodynia by relieving knee joint disability and pain. Taken together, LOXL2 deletion in mice knee promotes the severity PTOA, similar to human OA, implying its potential as a therapeutic candidate for human PTOA.
——————————————————————————————————————————–
Modified Medial Meniscectomy (MMM) Model to Assess Knee Joint Pain, Structure and Function in Post-Traumatic Osteoarthritis in Mouse. Article Link

Mechanical, physiological, and biochemical changes contribute to post-traumatic osteoarthritis (PTOA). However, specific mouse models that are highly reproducible, less invasive, and easy to perform are lacking, hampering our understanding of PTOA pain, function, and degeneration. This limitation hinders the progress of evaluating PTOA-related studies for diagnostic and therapeutic applications. The goal of this study was to establish a highly reproducible and efficient surgical model for PTOA. We developed a modified medial meniscectomy (MMM) model that demonstrated high reproducibility and applicability in assessing pain, as well as in conducting structural and functional analyses. The MMM model features distinct differences in the execution of transection of the medial meniscus on the lateral side and includes a smaller incision, which enhances reproducibility and is beneficial for studying pain, structure, and function. Consequently, this MMM model can be used for future mechanistic and therapeutic interventions.
