Engineering the Blind to See
Vision is one of the most impressive functions of the human brain. It interprets nothing but electromagnetic waves and paints a glorious picture of our daily existence from the scattered chaotic sea of intertwining light waves that we call home. Many see their vision deteriorate and the world blur as time goes on and these problems can be corrected by optometry, but blindness comes on like a relentless infidel for more than two million people worldwide in the form of retinitis pigmentosa (RP). RP is a heritible genetic disorder that leads to degeneration and loss of function in the retina’s photoreceptor cells, and can lead to full blindness in a matter of years. There is no cure or treatment for RP, but new research may change that very soon.
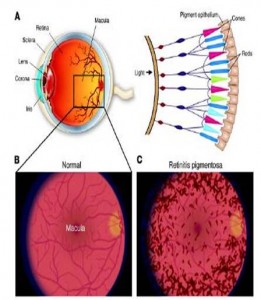
TOP: Macroscopic human eye (left) and rod and cone cell arrangement on the retina (right). BOTTOM: Normal human retina (left) and human retina with retinitis pigmentosa (right)/(Source: Medical-Look.com)
Vision starts in the photoreceptor when light activates rhodopsin, a G-protein coupled receptor pigment consisting of light sensitive opsin and retinal. Light triggers a conformational change in retinal that kick starts a G-coupled visual cascade and the flow of visual information to the brain. In retinitis pigmentosa, rhodopsins in the photoreceptors become insensitive to light starting in the rod cells, and blindness sets in gradually. Rod cells are used in low light and deteriorate first, leading to night blindness, and dysfunction in cone cells used for color vision and acuity sets in until full blindness plagues the individual. Fortunately, a team of French scientists has investigated this degradation and has found a way to combat it by reactivating the photoreceptor cells through genetics. Their study was featured in the July 23rd issue of Science.
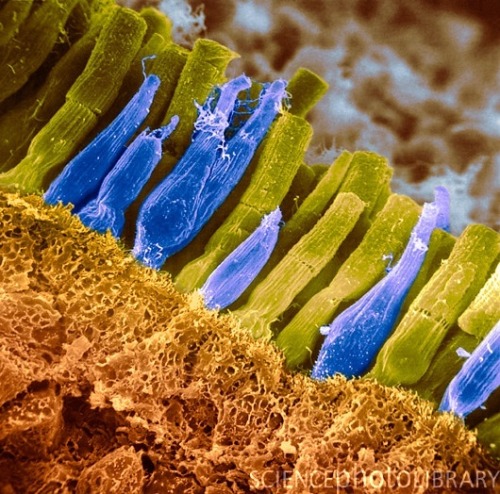
Artificially colored micrograph image of retinal rods (yellow-green) and cones (blue). (Credit: Science Photo Library @ sciencephoto.com)
The scientists isolated an achaebacterial rhodopsin analog called halorhodopsin that functions in the yellow and green wavelength range. They then introduced a halorhodopsin encoding gene into retinitis pigmentosa model mice via a viral vector and also created a control group. In their experiments, it was found that both slow and fast degrading retinal cells in the experimental mice regained their light sensitivity in response to integration of halorhodopsin into their insensitive photoreceptor cells. Electrical responses were recorded from ganglion cells (the third tier cell in the visual cascade) and healthy photoreceptor spikes were observed in response to light stimulation. Most importantly, lateral inhibition (the mechanism by which the brain discriminates edges of objects) was fully preserved, while mono-directional movement was retained. Halorhodopsin mice also performed significantly better than the control RP mice in a battery of visually guided tasks, demonstrating that their photoreceptors had been successfully resensitized by halorhodopsin integration. The scientists also tested the resensitizing ability of halorhodopsin on cultured human retinal cells. They were successful in integrating halorhodopsin into human cells via viral vectors, but could not conduct any clinical trials. However, photoreceptors expressing halorhodopsin demonstrated photocurrents and photovoltages that would be adequate to restore human vision.

Theoretical device that allows a halorhodopsin treated patient to see by projecting patterned light onto the eyes derived from camera input. (Credit: Y. Greenman/Science)
This opens the door to treatment of retinitis pigmentosa in the genes – the same place where it starts. Although halorhodopsin therapy will not fully restore all wavelengths in human vision, it can still serve as a tool to bring restore vision in the blind through optical devices. For example, an RP patient could be treated with halorhodopsin gene thearpy, then outfitted with a device that images the visual field and translates it into halorhodopsin recognizable wavelengths. This light mosaic is then projected onto the patient’s eyes and the can “see” what is in front of them. The supplied image of the device is from a perspective article in the beginning of the current issue of Science.
View the full text Science article here (HTML) or here (PDF) and the perspective piece about the article here. Be sure to discuss in the comments!
Sources:
Seeing the Light of Day – Science (Perspective)
Genetic Reactivation of Cone Photoreceptors Restores Responses in Retinitis Pigmentosa – Science (Research Article)
August 4, 2010
Great reporting, Frank!
I don’t understand one thing though – why would the treatments require an optical device to translate the light wavelengths of a visual field into ones that halorhodopsin “understands”? Wouldn’t the halorhodopsin do the translating itself?
As you say, halorhodopsin functions in the green and yellow wavelength range, but one has to keep in mind that (usually) the protein responds to a wide range of wavelengths, where green (for example) is just the one that activates it the best. So, for example, if our halorhodopsin is exposed to red light, it will still respond, but the perception would be of some shade of green/yellow instead of red.
This is just speculation – I don’t have much background in visual physiology. Thoughts?
August 4, 2010
Thanks, Greg…I hope to shed some light on your confusion (pun intended).
From what I understand, although halorhodopsin can still function within the frequencies of visible light, quality of perception would be greatly reduced when perceiving wavelengths outside the cells’ optimal range. For example, when receiving red light, the object emitting red wavelengths will appear untrue in the patient’s perception because their halorhodopsin cells’ respond to only the yellow/green portions of the red wavelength. This is where the optical device comes in – the device would capture images of the visual field in real time and translate them to the appropriate yellow and green wavelengths in the interest of mimicking true saturation and brightness perception for the patient.
Remember, we are not dealing with normal human photoreceptors anymore once they have been treated with AAV-Halorhodopsin. The LED glasses may be able to shine a mesh of wavelengths on the eyes of the patient that causes perception of other colors based on rates of hyperpolarization for given hues of yellow and green light. Think of a traffic light: because the patient’s halorhodopsin eyes mostly perceive yellow and green, they may not be able to discriminate the red color properly and perceive it inadequately, but the device will maintain the brightness and saturation of the red light so they can react appropriately.
Those are my thoughts, hope they make sense!