By ajun92
The Polygenetic Basis of Mutations in the Development of Schizophrenia

Schizophrenia is a mental disorder often presented in patients by abnormal thought processes, impaired emotional responses, and negative symptoms. As a chronic disorder that affects ~1% of all people, schizophrenia can be have debilitating effects on patients, especially on their social lives. Due to the lack of knowledge on its pathophysiology and also the heterogeneity of the symptoms, it has been increasingly important to understand the genetics of schizophrenia.
Due to the marked reduction in fecundity seen in schizophrenic patients, the high heritability of the disorder pointed to the possibility that genetic alleles that were risk factors might occur as de novo mutations. Previous exome sequencing studies showed no promising results, but the inconclusive results were likely due to small sample size and a narrow focus on target genes. Two recent studies, the largest of their kind, gathered data from nearly 7000 people (nearly 3500 patients) from Sweden and Bulgaria, and showed that genetic effects on schizophrenia seemed to be very complex. Specifically, both papers published in Nature reflected on the implication of genetic mutations in clusters of specific proteins that governed signaling networks dealing with learning and memory. The studies identified the presence of de novo mutations, often nonsense mutations, notably in genes related to the PSD (post-synaptic density of dendrites), the calcium channels, the postsynaptic ARC complex, and the NMDA receptors.
More
Why do we need sleep?
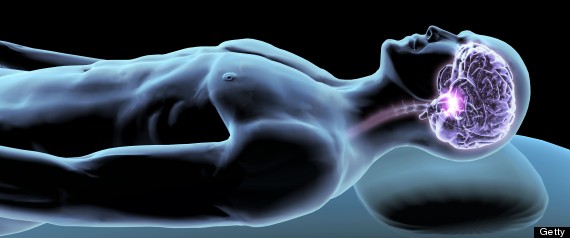
We know from everyday life that, at some point, we need to sleep. In fact, extended sleep deprivation can lead to death. Despite the amount of sleep research that has been conducted, none have been able to clearly reason out the essential function of sleep. However, recently, a promising study by Dr. Nedergaard showed that sleep functions in clearing neurotoxic waste from the brain of mice. In effect, without sleep, these toxins would build up and cause problems for the body.
Specifically, the study looked at what is known as the glymphatic system. Because our central nervous system lacks a lymphatic system which is in our peripheral system, the glymphatic clearance pathway is the primary way in which our brain can "clear" the cerebrospinal fluid (CSF) and interstital fluid (ISF) of the brain parenchyma. This clearance includes functions of getting rid of wastes, soluble proteins, and even controlling the volume of fluid. Interestingly, the Nedergaard study showed that this clearance system works faster when mice were asleep--in other words, the exchange rates of CSF and ISF increased during sleep. In addition, they were able to show that surrounding cells in the brain would shrink in size to allow more efficient clearance.
Research Spotlight on Nobel Laureate Thomas C. Südhof

Thomas C. Südhof
For those interested in scientific research, the Nobel Prize is an esteemed award, recognizing one’s work and dedication. However, many student researchers are often disillusioned about the kind of work that is put in to receive such high praise. More often than not, these individuals did not suddenly discover something new. To be sure, their findings are the fruits of years of research and passion in the field of science. As a great model of dedication, recent Nobel laureate Thomas Südhof provided incredible insight to our understanding of the human brain communication. But, once again, his work was in the basic-science field, on a topic which, in fact, we learn about in our introductory neuroscience courses. In this brief article, I will outline the most recent findings from Dr. Südhof’s lab in hopes of showing aspiring researchers that continued diligence and passion for learning is most important.
Neurons communicate with each other by a signaling process mediated by what is known as action potentials. Changes in concentration and electrical gradients cause the action potentials to fire down the neuron, until it reaches the synaptic bouton. It is here that one neuron forms a synapse with another neuron. The synapse is the site at which communication is happening. But, for the most part, two neurons are not physically connected, so how does communication happen? In a process called neurotransmission, when the action potential reaches the end of the neuron, an influx of Ca2+ ions cause vesicles to release certain chemicals out of the neuron, as a signal to the next. These vesicles are like little packets of neurotransmitter chemicals. Herein lies the question that Dr. Südhof sought to illuminate: mechanistically, how do these vesicles actually release the chemicals?