Tagged: Medicaid
Can Single-Payer Really Happen?
Healthcare is on the frontlines of legislative debates— the U.S. has the most expensive care and reports the poorest outcomes of all rich democracies (RDs). Several states have proposed legislation to innovate healthcare access and to safeguard against the destruction of the ACA. One “old idea” that recently gained momentum is single-payer, and for New York (NY), it may become a reality, with its fate resting with the new legislature. To date, only one state has briefly “experimented” with single-payer – Vermont, and it failed due to gross underestimation of its costs. In NY, a landmark study that evaluates the viability of NY’s single-payer bill, known as the New York Health Act (NYHA), conducted by RAND, detailed benefits and setbacks of the proposed legislation, and public reaction was mixed. Despite the not so encouraging findings, lessons can still be learned from this report.
Single-payer, coined as “Medicare for all”, is a health insurance system in which a single public agency organizes healthcare financing, ideally covering all types of essential healthcare services. Delivery of care itself, however, would remain largely private in a single-payer system.
Proposals for single-payer in the U.S. are not new. The earliest version came in 1943 by Senators Robert Wagner (D-New York), James Murray (D-Montana), and Representative John Dingell, Sr. (D-Michigan), known as the Wagner-Murray-Dingell Bill (and subsequently endorsed by President Truman in 1945). The post-World War II bill proposed funding health care through payroll and income taxes. The bill became entangled with the Cold War , was vilified as “socialized medicine” by its opponents, and was discarded. The idea was revived in the 1950s, when it was nearly impossible for an aging population to get private health insurance. The elderly advocated for subsidized coverage since they are no longer able to afford their care, and hospitals advocated for it to ensure that the healthcare services they provide were paid for. The result: Medicare was enacted in 1965— the first form of single-payer insurance in the U.S.
 Single-payer is gaining popularity once again. According to a Reuters poll, 70% of Americans support some form of single-payer coverage. Why? First, with the implementation of the ACA, there was a national momentum for states to expand their healthcare coverage. The health exchange created by the ACA made coverage accessible for many middle-income families and individuals. On the Medicaid side, progressive states elected to expand their eligibility coverage for individuals earning up to 138% of the federal poverty level in exchange for a 50% match in federal subsidies, a benefit many states enjoy. The most appealing provision of all is the mandated coverage of pre-existing conditions. With the all Republican take-over of the federal government in 2016, many Americans worried about what would happen to their coverage. Over the next two years, the ACA underwent congressional budget cuts, but despite efforts by Congress and President Trump, the ACA has grown in popularity with the general public.
Single-payer is gaining popularity once again. According to a Reuters poll, 70% of Americans support some form of single-payer coverage. Why? First, with the implementation of the ACA, there was a national momentum for states to expand their healthcare coverage. The health exchange created by the ACA made coverage accessible for many middle-income families and individuals. On the Medicaid side, progressive states elected to expand their eligibility coverage for individuals earning up to 138% of the federal poverty level in exchange for a 50% match in federal subsidies, a benefit many states enjoy. The most appealing provision of all is the mandated coverage of pre-existing conditions. With the all Republican take-over of the federal government in 2016, many Americans worried about what would happen to their coverage. Over the next two years, the ACA underwent congressional budget cuts, but despite efforts by Congress and President Trump, the ACA has grown in popularity with the general public.
In NY, the concept of single-payer was first introduced in 1992 by Assembly member Richard Gottfried (NY-D). The goal of NYHA is to provide universal insurance coverage with no cost-sharing for New Yorkers, regardless of legal status, and would cover almost all comprehensive services. Bill proponents expect increased access to care and reduced costs by removing high administrative overhead costs and reducing unjustifiably high prescription drug costs. Much like the Wagner-Murray-Dingell Bill, NYHA would be funded through payroll and income taxes. Since 2015, the NYHA has passed the Assembly floor four times. Although 31 state senators co-signed the bill, it has been stopped in the Senate by just one vote. This may now change with the Democrats taking back the Senate majority, although the cost may be a deterrent.
Despite the national and legislative enthusiasm, New Yorkers have been skeptical of single-payer reform. According to a 2018 Mercury Public Affairs poll, only 33% support the bill. Over 60%, however, said they would support increased subsidies to assist low and middle-income families. Why the opposition? The number one reason of 66% polled: taxes would pose a high burden.
The RAND study assessed “near-term” and “long-term” impacts of the bill. Overall, it found that single-payer would be viable, but with big caveats. The system would expand health care access, all while generating an estimated $15 billion in net savings (3.1%) on healthcare costs by 2031. Still, near-term are where the problems lie. From the political side, this would require the federal government to issue a waiver to redirect all federal and state funds to NYHA. Just weeks prior to this report, the Centers for Medicare & Medicaid Services called California’s similar proposal “unworkable” and indicated similar waivers would not be approved. On the fiscal side, health care reform comes with a steep price: $139 billion in additional state tax revenue would be needed by 2022, that is 156% more than what is currently being collected. This amount would be amassed through payroll and income tax that would supplant the employer contribution and premiums and out-of-pocket costs. RAND applied a generic tax schedule based on three income brackets. For low-income families, they would be taxed 6.1% of their payroll income and 6.2% for non-payroll income by 2022. For middle-income families, the rates would range from 12.2% to 12.4%, and for high-income, their tax rate would increase up to 18.3% – nearly three times of what they are paying now. Moreover, Medicaid and Essential Plan (i.e. the NY Health Exchange) enrollees would pay more to get healthcare coverage. Assembly member Gottfried praised the study and suggested that they can adjust taxes accordingly so that high income families would pay more in taxes in order to help low and middle-income families afford their care. These tax hikes would exceed the combined costs of what New Yorkers are currently paying in taxes and healthcare benefits—explaining the bill’s unpopularity. Using the RAND report as a guide, it is likely that the state legislature will explore mechanisms to help finance their proposal in the upcoming session.
While single-payer hasn’t had much luck in the U.S., universal care payment methods, including single-payer, have been successful in other RDs. Regardless of each RD’s financing method, there is one consistent feature of success: national political will to implement it. Imagine if the politics of the cold war did not interfere with establishing a national health insurance plan? Would it have been possible to implement a streamlined and efficient plan? If our culture would have capitalized on the Medicare momentum, would we accept a collective sense of community regarding our healthcare? Vermont tried to implement single-payer with little success due to gross budget underestimations and faint national support. The RAND report sheds light on the cost of single-payer and suggests that there needs to be federal political will to support it. Let these findings and other evidence guide lawmakers as the search for a modest solution continues. Perhaps Wagner’s vision may still be a solution.
Sarah Zahakos is working toward a PhD in Health Law, Policy & Management at the Boston University School of Public Health.
AHRQ T32 Research FellowTraining in Health Services Research for Vulnerable PopulationsGrant # 2T32HS022242
How State Medicaid Reimbursements are Limiting Hepatitis C Coverage and Solutions
Hepatitis C has rapidly become a major public health problem, and accessibility to new and affordable treatments has been highly sought after. Hepatitis C virus (HCV) can be transmitted by blood transfusions or contaminated needles; although no symptoms are present at the outset, chronic Hepatitis C infection can give rise to liver complications such as cirrhosis or liver cancer. As the leading infectious killer in the U.S., HCV chronically infects 2.7 million to 3.5 million people in the U.S. Since first coming onto the U.S. market in in 2013, Sovaldi has been one of the few, if only, medications to successfully treat Hepatitis C at a cure rate of over 90% after 12 weeks of treatment, with few side effects. Sovaldi holds considerable exclusivity due to its novelty. As such, Sovaldi costs $1,000 per pill, leading to a costly $84,000 for a 12-week treatment regimen. In contrast, Sovaldi costs less than $1 per pill.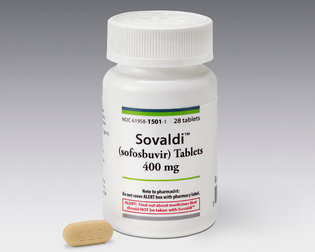
Sovaldi pricing has received nationwide attention; to the point of rare bi-partisan partnership taking action. On July 11, 2014, Senate Finance Committee Ranking Member Ron Wyden (D-Oregon) and senior Committee Member Chuck Grassley (R-Iowa) requested information on Gilead Sciences’ pricing tactics that have negatively affected public payers’ access to Sovaldi. The lawmakers deemed the investigation pertinent since the medication raises “serious questions about the extent to which the market for this drug is operating efficiently and rationally,” and Sovaldi’s price “appears to be higher than expected given the costs of development and production and the steep discounts offered in other countries.” After an 18-month investigation and hearing, Congress released an extensive report on December 1, 2015, detailing Gilead Sciences’ pricing, marketing, and development mechanisms on Sovaldi and Harvoni.
Among other conclusions explaining the price variation, the report determined that Gilead did not easily provide access to states’ Medicaid programs. Medicaid programs spent $1.3 billion on Sovaldi, before rebates, in 2014 alone, yet less than 2.4 percent of Medicaid-eligible patients with HCV received treatment. In fact, many states have resorted to restricting access to covering a limited number of Medicaid patients. Some states, such as New Mexico, limit Medicaid coverage to the sickest patients, or those who have pre-existing liver damage. These states require healthcare providers to “perform risky liver biopsies on patients to prove how sick they are, or wait until patients have late-stage liver disease before they can be eligible for coverage.”
A recent study examined how consistently state Medicaid programs abided by recommendations made by the American Association for the Study of Liver Disease and the Infectious Diseases Society of America on treating, managing, and preventing HCV. Out of the 42 states (including the District of Columbia) that had publicly available Medicaid reimbursement criteria, 74% restricted access to Solvaldi to those with advanced fibrosis or cirrhosis, which occurs at stage F4 of the disease. Furthermore, a majority of states limit Medicaid reimbursements to patients who have abstained from drug and alcohol use for a certain period of time, even those who have undergone opioid substitution therapy. From these findings, the study concluded that current state Medicaid reimbursements may violate federal Medicaid law, which provides that state Medicaid programs plans must include drugs manufactured by pharmaceutical companies that have negotiated rebate plans with the Secretary of Health and Human Services, except for those under the restrictive lists of drugs.
Some states have rearranged their Medicaid program policies by forming a pool with other states and purchasing alternative treatments exclusively in an effort to force coverage away from Solvaldi. As of January 2015, Missouri and 24 other states successfully reached a negotiation with AbbVie to secure an extra 20 to 30 percent rebate system for their Medicaid patients. However, these negotiations place restrictions on Medicaid enrollees, who are required to stay sober for 90 days before beginning the Viekira Pak treatment. Although Missouri anticipates to save $4.2 billion from these rebates, the lag time of over six months for calculating rebates into the state budget means it will be too soon to determine the overall cost savings on Medicaid spending. The Missouri state department also plans to provide Solvaldi to an estimated 15 to 20% of HCV patients who cannot be effectively treated by Viekira Pak, which implies that the medication cannot clinically treat all people infected by HCV. Exclusive Medicaid negotiations with alternative treatments could lead to unintended leftover costs from providing for patients whom Viekira Pak is not a valuable option.
Recent litigation on the state level has addressed issues of Medicaid coverage of Sovaldi. In B.E. v. Teeter, Washington Medicaid enrollees, who were HCV patients that did not receive DAA medication, brought a class action suit against the Washington State Health Care Authority (“WHCA”), under claims of violating the Medicaid Act for categorically excluding them from “medically necessary” drugs. The federal district court sided with the plaintiffs’ argument, granting their motion for preliminary injunction, given that the plaintiffs had satisfied all the factors necessary to warrant such a remedy. In doing so, the court determined that the plaintiffs’ evidence “will likely establish that the WHCA is failing to follow its own definition of medical necessity by refusing to provide DAAs to monoinfected enrollees with a F0-F2 score and offering only “monitoring” in lieu of this breakthrough treatment.”
This decision significantly marked the first time that a federal court deemed restrictions to Hepatitis C state Medicaid programs as illegal, and thus could provide a precedent for other states to follow suit. Consumers from California and, again Washington, have also recently filed suits against private insurance companies, such as Anthem Blue Cross and Group Health Cooperative. Since these lawsuits have involved gathering a class of injured plaintiffs, however, issues of class certification under Rule 23(a) and 23(b)(2) will need to be resolved, as they were in B.E. v. Teeter. As such, law suits filed on behalf of a consumer class may not be the most efficient resolution, since time constraints and litigation costs could prolong the desired remedy, if the court chooses to grant it.
Massachusetts employs a fee-for-service program for distributing Solvaldi. As such, the state has relatively unrestricted access to Sovaldi compared to other states; yet, only an estimated 1,075 members have been approved for treatment regimens among the 7,658 members living with HCV. In an unprecedented move, on January 2016, Massachusetts Attorney General Maura Healey issued a letter warning to sue Gilead Sciences for potentially violating unfair trade practice under section 2 of chapter 93A of the Mass. Gen. Laws. However, the lawsuit may not contain the merits required to bring an action under consumer protection law. The AG eventually spent months negotiating with Gilead Sciences and recently, on June 30, 2016, reached a new drug rebate program to provide unrestricted coverage to MassHealth patients in need of Hepatitis C treatment. Since the MassHealth rebate program was recently implemented on last August 1st, the effectiveness of the pricing solution will need to be monitored further to determine whether Medicaid coverage of Solvaldi is expanded to offer more treatments to those in need.
 Monica Chou anticipates graduating from Boston University School of Law in May 2018 and plans to practice health law.
Monica Chou anticipates graduating from Boston University School of Law in May 2018 and plans to practice health law.
With Obamacare in peril, the Governors Speak Out
Before the Affordable Care Act (ACA), I did not have health insurance. My home state Florida did not mandate health insurance coverage for residents and my undergraduate university did not require me to have health insurance. In essence, I was a typical American in my early 20s. I did not think I needed health insurance, was not required to carry it, and could not afford it. Cost was the greatest factor: I did not have any income and Florida did not expand Medicaid. To my surprise, the ACA allowed me to have affordable health insurance for the first time as an adult. Now Congress is contemplating major changes to the ACA (or commonly called Obamacare), causing some governors, such as Charlie Baker (R-Massachusetts), to weigh in on the proposals. Despite the recent successful House repeal and replacement of the ACA, the Senate is now struggling to find a path forward under the budget reconciliation rules.
Just a few months ago, it looked as though changes to the ACA were inevitable given the unified Republican control of Congress and the Administration. Despite seven years of discussion of repeal, and more recently the repeal and replace vote, the future is still uncertain. Members of Congress who attended town hall meetings during the 4th of July break heard from many constituents who are deeply concerned with loss of coverage. Former House Speaker John Boehner recently said that Congress would not repeal and replace, but instead “fix” Obamacare. And now, the Senate is spinning its wheels.
The much-awaited House bill, the American Healthcare Act (the “AHCA”), was the first attempt at replacing the ACA. The AHCA would repeal tax penalties for people without health insurance, reduce federal insurance standards, cut subsidies for buying private insurance and establish new limits on spending for Medicaid. In their first attempt, Republicans failed to get their bill to reach the House floor for a vote. In spite of this defeat, the Trump Administration placed renewed pressure on Congress to revise the ACA. Ultimately, the House was able to pass the bill in its second try by adding $8 billion to help cover insurance costs for people with pre-existing conditions. However, the Senate cannot pass the House bill because Majority Leader McConnell must accommodate Republican senators from states that have expanded Medicaid under the ACA. These senators, with a statewide constituency, must consider what their state governors have to say on revisions to the ACA.
Congress' ACA replacement process included a request to the 50 governors for information. In response, Massachusetts Governor Baker sent a letter on the ACA’s impact on Massachusetts. Some commentators believe Governor Baker’s letter could carry extra weight because of his Republican party affiliation and his past work experience as chief executive of Harvard Pilgrim Health Care gives his suggestions and concerns greater authority.
In his letter, Governor Baker discussed the importance of the health sector to the Massachusetts economy; $19.77 billion, making it one of the leading industries in the state. Governor Baker also noted that the ACA was modeled after the Massachusetts system, which was intended to provide close to universal coverage for residents. Massachusetts has the highest percentage of insured residents in the U.S.— 96.4%. Just under 60% of the insured are covered through the employer-sponsored insurance market.
Governor Baker argued that lawmakers should not repeal the ACA, but revise it. One area in need of repair is the ability of individuals with employer provided insurance to switch to tax-payer subsidized health insurance; something half a million Massachusetts residents have done since 2011. As a result, Medicaid now accounts for close to 40% of the state’s budget. Since 2012, the percentage of Massachusetts residents on commercial insurance decreased by 7% while Medicaid enrollment increased by 7% and now insures 28% of the population. The original Massachusetts program did not allow this transfer, but the State was forced to comply with the introduction of the ACA. Now, this particular aspect of the ACA was straining the Massachusetts system and needed to be revised. Although Governor Baker offered reforms, he argued for maintaining several aspects of the law, such as the mandate requiring all residents to carry health insurance, which would allow stability within high-risk pools for insuring people who are sick.
The Governor continues to push the goal of universal health care coverage, but recognized such a goal was in jeopardy because of certain Congressional proposals. For instance, the letter expressed concern over a shift to block grants for Medicaid funding to the states. The Governor argues that a shift to block grants (or “per capita caps for Medicaid) would “remove flexibility from states” as the result of lower federal funding. Under current law, the federal government and state governments share in the financing and administration of Medicaid. According to the Congressional Budget Office, states typically pay health care providers for services to enrollees, and the federal government reimburses states for a percentage of their expenditures. Furthermore, all federal reimbursement for medical services is “open-ended” in other words, if a state spends more because enrollment increases or costs per enrollee rise, additional the federal government matches. Currently, Massachusetts is a 50/50 state, meaning that the federal government and Massachusetts divide the cost of providing health care for Medicaid recipients.
Despite Governor Baker position, the House passed AHCA creates a per capita-based cap on Medicaid payments for medical assistance. The per-capita caps would establish a limit on the amount of reimbursement the federal government provides to states. For instance, if a state spent more than the federally established limit on reimbursements, the federal government would not match the additional costs. The AHCA would punish Massachusetts low income residents and threaten the stability of the MassHealth system. Consequently, the changes to federal grants of funds could impact the Commonwealth’s goal of universal healthcare.
Today, Governor Baker—joined with nine other governors, including Gov. Sandoval (R-Nevada) and Gov. Kasich (R-Ohio) —sent another letter to the Senate urging it to correct the ACA's weaknesses without repealing the law or gutting Medicaid. The Governors wrote, "lasting reforms can only be achieved in an open, bipartisan fashion." The governors also called on the Senate to heed U.S. Sen. John McCain's, R-Arizona, impassioned plea to return to "regular order" and not continue the recent practice of hyper partisanship.
The governors are speaking; the question remains whether Congress is listening.
 Juan Garay graduated from Boston University School of Law in 2017.
Juan Garay graduated from Boston University School of Law in 2017.


